Noblyst®: Selective where it counts
Evonik’s catalysts for selective hydrogenation are a classic success story. Developed as custom catalysts for specific processes several decades ago, they have since established themselves as global industry standards for vital industrial & petrochemical applications evolving significantly in the meantime.
The Noblyst® brand precious metal fixed bed catalysts have been developed by Evonik over the last three decades as tailored catalysts for specific applications, such as the production of propylene or phenol. Considering this long history, it speaks volumes to the quality of the catalysts, that they are still being utilized as state of the art technology around the world.
C3/C4-Chemistry
For the processing of C3 and C4 hydrocarbon streams in liquid phase process types, highly active palladium on alumina catalysts such as Noblyst® H14171 and Noblyst® H14271 are ideal. These catalysts can be used for either complete hydrogenation of double or triple bonds or selective hydrogenation, keeping one unsaturated bond intact. The related processes are the complete saturation process (CSP) and the selective hydrogenation process (SHP). Due to their high activity and selectivity the Noblyst® catalysts permit operation at low temperatures and high space velocities.
Noblyst® catalysts are applied in the production of propylene from propane. After the dehydrogenation unit, the separated propylene stream contains trace amounts of acetylenes and diolefins. These are in turn eliminated in an SHP unit to avoid side reactions in downstream processes. In the production of methyl tertiary butyl ether (MTBE), various olefins and diolefins, remaining after the separation of MTBE are finally saturated to butanes in a complete saturation process (CSP) unit. These are examples where over the years major progress has been made in enhancing the catalysts’ efficiency.
Dehydrogenation Process for Propylene Production
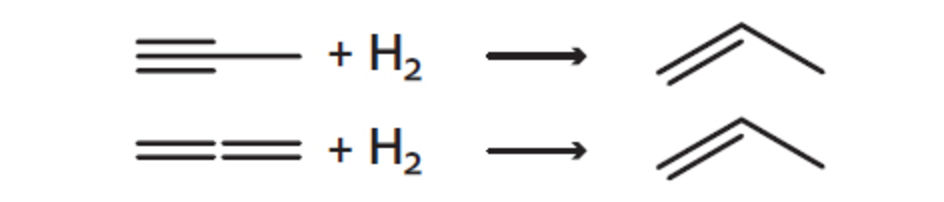
This catalytic process produces propylene from propane. After the dehydrogenation unit the separated propylene stream contains methylacetylene and propadiene which are converted to the desired product propylene in an SHP unit.
Process for MTBE Production
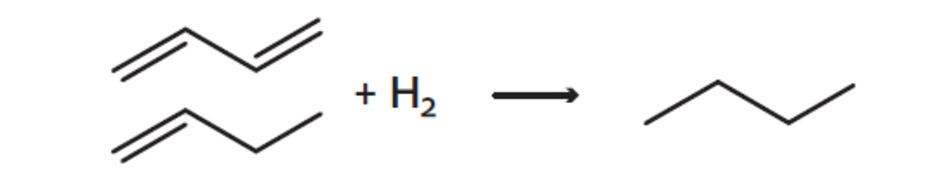
Isobutylene is used to produce methyl tertiary butyl ether (MTBE). n-Olefins and diolefins remaining after the separation of MTBE are finally saturated to butanes in a CSP unit which can be fed back into the isomerization process.
Alpha-Methylstyrene (AMS)
Typically, the production of phenol and acetone is based on the Hock process starting from cumene. One of the byproducts of this process is alpha-methylstyrene (AMS), which can be separated and used as a co-polymer, for example in Acrylonitrile butadiene styrene (ABS) production. However, much more efficient is the selective hydrogenation to cumene using Noblyst® H14108 and refeed into the oxidation step.
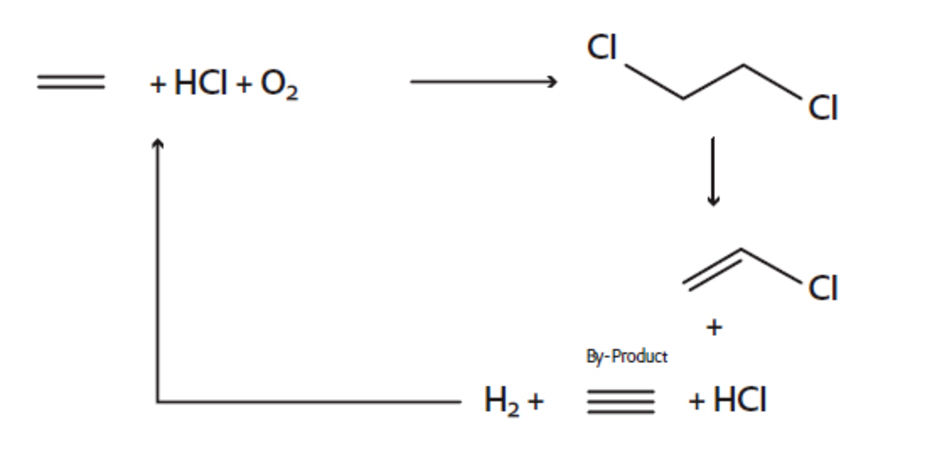
Vinyl Chloride Monomer (VCM)
Vinyl chloride monomer (VCM) is the base material for the production of polyvinyl chloride (PVC), which is one of the most widely used polymers, employed in the construction industry for flooring, cable insulation and window profiles. 40 years of experience in this application have made Evonik the frontrunner for the catalytic hydrogenation process step in the production of VCM. This is where Evonik’s catalyst Noblyst® E39 series comes into its own.
In addition to the main product VCM, hydrochloric acid (HCl) and traces of acetylene are formed that remain in the HCl recycle stream. In a separate selective hydrogenation reactor, the unwanted acetylene is hydrogenated to ethylene, which can then be recycled into the oxychlorination process for conversion to ethylene dichloride (EDC). In this way catalysts of Evonik provide high added value in a resource-conserving and environmentally-friendly VCM production.
For high space velocities above 4000 1/h we recommend the high performance grade Noblyst® E39K.